
Superior flap tuck in mastopexy—a novel technique to promote lift and volume
Introduction
Breast ptosis is a complaint that presents to plastic surgeons globally (1). Its pathogenesis is well described, yet its surgical management can be challenging. The triad of progressive dermal laxity, inferior displacement of the nipple-areolar complex (NAC) and loss of superior pole fullness (bottoming out) leads to an aged breast appearance. From the 1900s onwards dozens of techniques involving skin incision and glandular resection to address these issues have been well described (2-6). These range from a multiple variations of inferior based pedicle flaps (6) to upper pole synthetic mesh integration (7) in an attempt to provide lasting volume. Hall-Findlay produced a large volume of positive outcomes using the vertical scar technique in the early 2000s yet the learning curve for some surgeons still dissuades them from its use (7). It is widely accepted that all techniques described suffer from their own drawbacks either through increased scar burden or bottoming out of final shape.
Native glandular tissue redistribution to correct loss of upper pole fullness is not a new technique (7). First described by Calderia in 1991 (8) the triple flap technique was designed to produce conification of the breast tissue to aid projection. Subsequently modified by Kelemen (8) the technique they describe involves shifting inferiorly based glandular tissue into a more cranial position behind the NAC. The superomedial NAC pedicle is “stacked” between the inferior flap pedicle and the nipple for further projection of the breast (9).
These commonly described inferiorly based glandular flaps however suffer from the drawbacks of eventual migration of the inferior breast tissue to its original position. Additionally, in our practice we have found inferiorly based flaps folded up lack volume in the most distal aspect reducing total tissue volume available directly behind the NAC. In this article we describe a novel superiorly based triple flap mastopexy technique via a vertical scar skin resection that maximizes projection via autologous breast tissue redistribution.
Case presentation
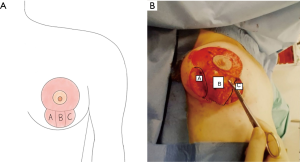
Skin markings are drawn preoperatively with the patient in a sitting position. The patient is marked as usual for mastopexy using the vertical technique. The breast meridian is marked, and the new nipple positions are set at the level of the inframammary fold (IMF). A 4-cm areolar diameter is planned, with the nipple kept on a superomedial pedicle and modified as required intraoperatively.
Surgery commences with an incision via a standard vertical scar pattern. The inferior boundary of the incision is kept above the IMF—an important structure to preserve for lasting breast architectural integrity. A superior pedicle is then outlined and de-epithelized as per standard reduction mastopexy approach. Following this the inferior glandular tissue is then divided into a triple flap design—A/B/C (Figure 1). A and C are cut down to fascia to isolate B similar to Lassus technique but without skin undermining (9). The medial (superiorly based) glandular flap B is dissected out, divided inferiorly then tucked back and secured to itself and the prepectoral fascia with 3.0 monocryl interrupted sutures (Figure 2), immediately posterior to the NAC. This location is similar to the placement position of a prosthesis if augmentation mastopexy were simultaneously being carried out but negates the need for such an intervention. The lateral flaps A + C are then brought together and secured over the top of A again with a 3.0 monocryl suture (Figure 3). Minimal dissection of flaps A + C is carried out. Hemostasis is ensured then the skin envelope is then closed with 3.0 monocryl deep dermal sutures. To address the dog ear at the terminal aspect of the vertical limb a modified deep dermal gathering suture is used which loops under the last stitch to gather redundant tissue.
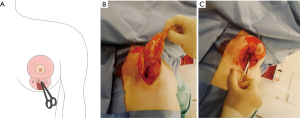
Final layer closure is then completed with a standard 3.0 monocryl subcutaneous. Glue and steristrips are used to cover the suture lines and the nipple. The patient is placed in a soft bra and left undisturbed until a wound check at day 7. No drains are used.
Comments
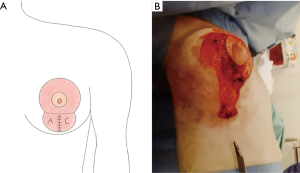
The senior author has moved to this technique as his standard approach for mastopexy surgery not requiring co-current reduction. We find that although the breast does, “settle”, into its final position approximately 6 months postoperatively NAC position and upper pole fullness is maintained (Figures 4,5). We believe this is due to the transposition of the superiorly based autologous tissue providing structured projection. To date nipple sensation has remained in all patients and no revisional surgery has been required.

The use of blocking sutures as originally described by Benelli (10) identified the crucial keystone area around the NAC supporting the mammary cone. It was initially felt that securing this area with a blocking suture could address inferior displacement of the complex and overall breast shape. With increasing ptosis and larger breasts however, this approach is rarely sufficient to produce lasting outcomes.
Redistribution of glandular breast tissue was thus identified as a further critical element in further correcting the factors that create an aged breast appearance, especially the loss of upper pole volume that occurs with time (11-13). Volume can be addressed with simultaneous augmentation (14) however this is not without risk, increasing the complexity of the surgery and reticence of some patients given the emerging risk of breast implant associated-anaplastic large cell lymphoma (BIA-ALCL) with textured implants can dissuade use. Thus, use of autologous tissue to address volume is the ideal, with multiple techniques as above already described. These techniques however all rely on stacking or redistribution of glandular tissue that is inferiorly based either in the breast or on the chest wall (8). The inherent issue with this is that although the tissue is secured with non-absorbable sutures, with time and the effects of gravity the glandular tissue migrates to its original position inferiorly.
The use of superiorly based glandular tissue redistribution as described in our technique reduces the risk of this. The anatomic union between the NAC and superiorly based medial flap B means that when the flap is tucked in and secured to the chest wall tissue migration is minimal. Additionally, as the flap is being folded posterior to the NAC rather than up to it from an inferior position (as in the stacked technique) glandular volume directly behind the NAC contributing to conification and projection is maximized. The described technique allows upper pole redistribution of native tissue preventing requirement for mesh or implant while minimizing scar burden in the skin envelope. It corrects ptosis while increasing the projection and apparent volume of the breast.
Although breast tissue redistribution is a recognized mastopexy technique we believe this is the first occasion that a superiorly based glandular flap has been described to produce augmentation of breast shape via a vertical scar. The senior author has found this technique produce a reliable and cosmetically appealing outcome for patients over the past 5 years.
Acknowledgments
Funding: None.
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/abs.2019.11.02). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in studies involving human participants were in accordance with the Declaration of Helsinki (as revised in 2013). Informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Vrebos J, Dupuis C. From a single vertical scar to vertical mammaplasty. From Louis Dartigues (1869-1940) to Claude Lassus (1933). Ann Chir Plast Esthet 2000;45:62-8. [PubMed]
- Aufricht G. Mammaplasty for pendulous breasts; empiric and geometric planning. Plast Reconstr Surg (1946) 1949;4:13-29. [Crossref] [PubMed]
- Peixoto G. Reduction mammaplasty: a personal technique. Plast Reconstr Surg 1980;65:217-26. [Crossref] [PubMed]
- Marconi F. The dermal pursestring suture: a new technique for a short inframammary scar in reduction mammaplasty and dermal mastopexy. Ann Plast Surg 1989;22:484-93; discussion 494. [Crossref] [PubMed]
- Parenteau JM, Regnault P. The Regnault "B" technique in mastopexy and breast reduction: a 12-year review. Aesthetic Plast Surg 1989;13:75-9. [Crossref] [PubMed]
- Marchac D, de Olarte G. Reduction mammaplasty and correction of ptosis with a short inframammary scar. Plast Reconstr Surg 1982;69:45-55. [Crossref] [PubMed]
- Hall-Findlay EJ. Vertical breast reduction. Semin Plast Surg 2004;18:211-24. [Crossref] [PubMed]
- Kelemen N, Kannan RY, Offer GJ. A stacked technique of mastopexy: volume redistribution mastopexy with inferior flap and superomedially based pedicle. Aesthetic Plast Surg 2013;37:349-53. [Crossref] [PubMed]
- Caldeira AM, Lucas A, Grigalek G. Mastoplasty: the triple-flap interposition technique. Aesthetic Plast Surg 1999;23:51-60. [Crossref] [PubMed]
- Lassus C. The Lassus vertical technique. Aesthet Surg J 2011;31:897-913. [Crossref] [PubMed]
- Graf R, Reis de Araujo LR, Rippel R, et al. Reduction mammaplasty and mastopexy using the vertical scar and thoracic wall flap technique. Aesthetic Plast Surg 2003;27:6-12. [Crossref] [PubMed]
- Hall-Findlay EJ. Pedicles in vertical breast reduction and mastopexy. Clin Plast Surg 2002;29:379-91. [Crossref] [PubMed]
- Hönig JF, Frey HP, Hasse FM, et al. Autoaugmentation mastopexy with an inferior-based pedicle. Aesthetic Plast Surg 2009;33:302-7. [Crossref] [PubMed]
- Benelli L. A new periareolar mammaplasty: the "round block" technique. Aesthetic Plast Surg 1990;14:93-100. [Crossref] [PubMed]
Cite this article as: McGarry KM, Johnston EN, Martin S, Khan K. Superior flap tuck in mastopexy—a novel technique to promote lift and volume. Ann Breast Surg 2019;3:24.


